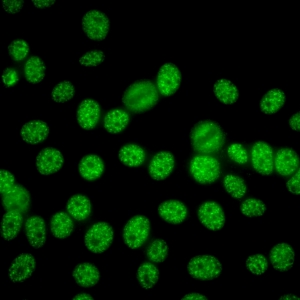
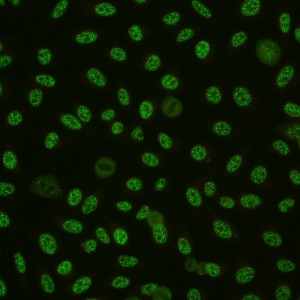
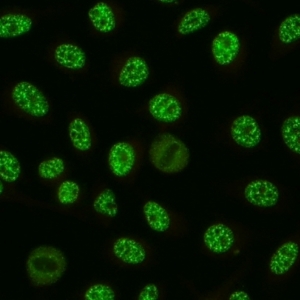
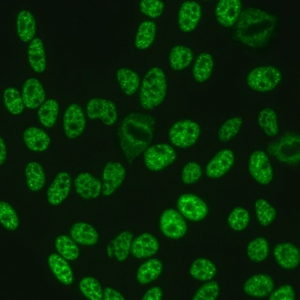
AC-5 - Large/Coarse Speckled
Descrição
Coarse speckles across all nucleoplasm. The nucleoli may be stained or not stained. Mitotic cells (metaphase, anaphase, and telophase) have the chromatin mass not stained. e.g. anti-Sm, anti-U1 RNP.
Associação Antigênica
hnRNP, U1RNP, Sm, RNA polymerase III.
Doença Associada
MCTD, SLE, SSc.
Relevância Clínica (Primeiro Nível)
Present to a varying degree in distinct SARD, in particular SLE, SSc, MCTD, SSc-AIM overlap syndrome, and UCTD (i.e, patients with rheumatic symptoms without a definite SARD diagnosis).
If SLE is clinically suspected, it is recommended to perform follow-up tests for anti-Sm and anti-U1RNP antibodies; these antigens are commonly included in the routine ENA profile; anti-Sm antibodies are included in the classification criteria for SLE.
If SSc is clinically suspected, it is recommended to perform a follow-up test for anti-RNApol III antibodies (e.g., SSc profile*); the anti-RNApol III antibodies are included in the classification criteria for SSc.
If MCTD is clinically suspected, it is recommended to perform a follow-up test for anti-U1RNP antibodies; the antigen is commonly included in the routine ENA profile; anti-U1RNP antibodies are included in the diagnostic criteria for MCTD.
If the SSc-AIM overlap syndrome is clinically suspected, it is recommended to perform follow-up tests for anti-U1RNP and anti-Ku antibodies; these antigens are included in the routine ENA profile (U1RNP), or in disease specific immunoassays (Ku, i.e., inflammatory myopathy profile and SSc profile)
In non-SARD individuals in the general population, the presence of the AC-5 pattern is not associated with the autoantigens mentioned above and most often concerns low antibody titers.
Relevância Clínica (Segundo Nível)
Occasionally, autoantibodies revealing the AC-5 pattern are reactive with RNP other than U1RNP, for instance U2RNP (associated with SSc-AIM overlap syndrome) or U11/U12RNP (associated with SSc); these autoantibodies can be detected by immunoprecipitation.
Specific immunoassays for these autoantibodies are currently not commercially available.
Referências
Betteridge Z, McHugh N. Myositis-specific autoantibodies: an important tool to support diagnosis of myositis. J Intern Med 2016;280:8–23.
Fretig N, Domsic RT, Rodriguez-Reyna T, et al. Anti-U11/U12 RNP antibodies in systemic sclerosis: a new serologic marker associated with pulmonary fibrosis. Arthritis Rheum 2009;61:958-65.
Mimori T, Hinterberger M, Pettersson I, et al. Autoantibodies to the U2 small nuclear ribonucleoprotein in a patient with scleroderma-polymyositis overlap syndrome. J Biol Chem 1984;259:560-5.
Satoh M, Fritzler MJ, Chan EKL. Antihistone and antispliceosomalantibodies. In: Lahita RG, Tsokos G, Buyon JP, et al, eds. Systemic lupus erythematosus. San Diego: Academic Press, 2011: 275–92.
Satoh M, Va?zquez-Del Mercado M, Chan EK. Clinical interpretation of antinuclear antibody tests in systemic rheumatic diseases. Mod Rheumatol 2009;19:219–28.
van den Hoogen F, Khanna D, Fransen J, et al. 2013 classification criteria for systemic sclerosis: an American College of rheumatology/European League against rheumatism collaborative initiative. Ann Rheum Dis 2013;72:1747–55.
Satoh M, Tanaka S, Ceribelli A, et al. A comprehensive overview on myositis-specific
antibodies: new and old biomarkers in idiopathic inflammatory myopathy. Clin RevAllergy Immunol 2017;52:1–19.
Sharp GC, Irvin WS, Tan EM, et al. Mixed connective tissue disease--an apparently
distinct rheumatic disease syndrome associated with a specific antibody to an
extractable nuclear antigen (ENA). Am J Med 1972;52:148–59.
Northway JD, Tan EM. Differentiation of antinuclear antibodies giving Speckled staining patterns in immunofluorescence. Clin Immunol Immunopathol 1972;1:140–54.
Petri M, Orbai AM, Alarco?n GS, et al. Derivation and validation of the systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012;64:2677–86.