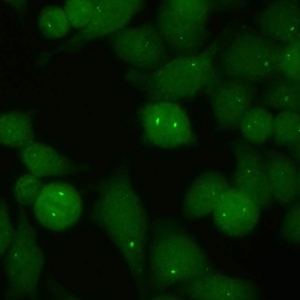
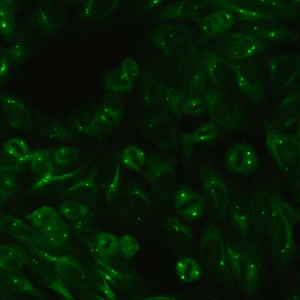
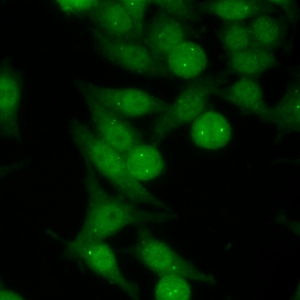
AC-24 - Centrosome
Descrição
Distinct centrioles (1-2/cell) in cytoplasm and at the poles of mitotic spindle.
Associação Antigênica
pericentrin, ninein, Cep250, Cep110
Doença Associada
Rare in SSc, Raynaud`s phenomenon, infections (viral and mycoplasma)
Relevância Clínica (Primeiro Nível)
The AC-24 pattern has low positive predictive value for any disease
Within the spectrum of the SARD, the AC-24 pattern
is found in patients with Raynaud’s phenomenon, localized scleroderma, SSc, SLE and RA, either alone or in combination with other SSc-associated antibodies;
102–105
Antigens recognized include ?-enolase, ?-enolase, ninein, Cep-250, Mob1, PCM-1/2, pericentrin; specific immunoassays for these autoantibodies are currently not commercially available
Relevância Clínica (Segundo Nível)
Possible association with infections; described in children with Mycoplasma pneumoniae infection
Possible association with malignancies; autoantibodies reacting with antigens in centrosomes are frequently found in sera of patients with breast cancer
Autoantibodies to ?-enolase, ?-enolase, Mob1, PCM-1/2, and pericentrin have been described in children with cerebellar ataxia after varicella infection; one patient with hyperthyroidism and vague muscle pain and one patient with Raynaud’s phenomenon, both without evidence of a SARD, had antibodies to ?-enolase and ?-enolase
With respect to autoantibodies to ninein and Cep-250, there is no apparent correlation between serum autoantibody reactivity and the clinical diagnosis; reported in RA and SLE
Most reports describe autoantibodies directly binding to specific antigens (i.e., antigen-specific immunoassays) and do not actually show correlations with the AC-24 pattern as such; specific immunoassays for these autoantibodies are currently not commercially available.
Referências
Takahashi T, Asano Y, Hirakawa M, et al. Linear scleroderma with prominent multiple lymphadenopathy followed by the development of polymyositis: a case report and review of published work. J Dermatol 2016;43:1224–7.
Howng SL, Chou AK, Lin CC, et al. Autoimmunity against hNinein, a human centrosomal protein, in patients with rheumatoid arthritis and systemic lupus erythematosus. Mol Med Rep 2011;4:825–30.
Hamaguchi Y, Matsushita T, Hasegawa M, et al. High incidence of pulmonary arterial hypertension in systemic sclerosis patients with anti-centriole autoantibodies. Mod Rheumatol 2015;25:798–801.
Gavanescu I, Vazquez-Abad D, McCauley J, et al. Centrosome proteins: a major class of autoantigens in scleroderma. J Clin Immunol 1999;19:166–71.
Fritzler MJ, Zhang M, Stinton LM, et al. Spectrum of centrosome autoantibodies in childhood varicella and post-varicella acute cerebellar ataxia. BMC Pediatr 2003;3.
Rattner JB, Martin L, Waisman DM, et al. Autoantibodies to the centrosome (centriole) react with determinants present in the glycolytic enzyme enolase. J Immunol 1991;146:2341–4.
Mack GJ, Rees J, Sandblom O, et al. Autoantibodies to a group of centrosomal proteins in human autoimmune sera reactive with the centrosome. Arthritis Rheum 1998;41:551–8.
Cimolai N, Mah D, Roland E. Anticentriolar autoantibodies in children with central nervous system manifestations of Mycoplasma pneumoniae infection. J Neurol Neurosurg Psychiatry 1994;57:638-9.
Madrid FF, Maroun MC, Olivero OA, et al. Autoantibodies in breast cancer sera are not epiphenomena and may participate in carcinogenesis. BMC Cancer 2015;15:407.
Maroun MC, Olivero O, Lipovich L, et al. Anti-centrosome antibodies in breast cancer are the expression of autoimmunity. Immunol Res 2014;60:339-47.
Fritzler MJ, Zhang M, Stinton LM, et al. Spectrum of centrosome autoantibodies in childhood varicella and post-varicella acute cerebellar ataxia. BMC Pediatr 2003;3:11.